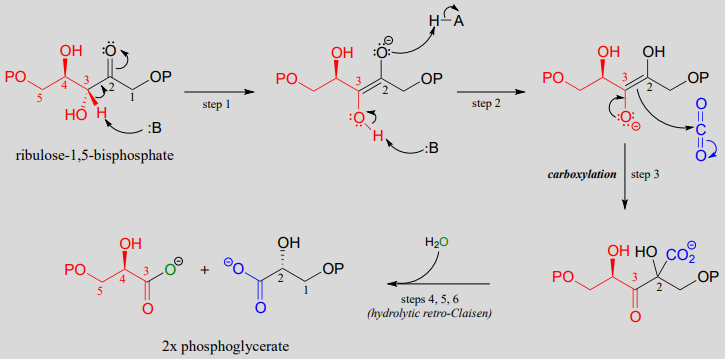
Imagine you're a tiny, super-enthusiastic chef, constantly looking for the perfect ingredient. That's kind of like a Grignard reagent. They're little chemical powerhouses, always ready to grab onto something exciting!
Our star chef today wants carbon dioxide (CO2), the same stuff we breathe out. To a Grignard reagent, CO2 is like a delicious, slightly sour candy they just have to get their hands on.
The Great CO2 Caper
So, how does this kitchen caper unfold? It's a bit like a very determined hug, but with atoms. The Grignard reagent, which we'll nickname "Griff" for short, is basically a carbon atom attached to a magnesium atom. Magnesium is naturally a little more positive, and the carbon is slightly negative.
Think of magnesium as Griff’s overbearing stage-parent constantly pushing Griff into situations. This slight imbalance is key to Griff's reactive personality.
Griff Meets CO2
Now, CO2 waltzes into the kitchen. It’s a linear molecule, with a carbon atom double-bonded to two oxygen atoms. Those oxygen atoms are electron hogs, making the carbon in CO2 a little bit electron-deficient. Perfect for Griff!
Griff, driven by magnesium's encouragement and his slight negative charge, lunges at the carbon atom in CO2. It's like a kid seeing ice cream on a hot day – irresistible! Griff grabs the CO2's carbon.
This isn't just a casual handshake; it's a full-on embrace! One of the double bonds between carbon and oxygen in CO2 breaks. It's the chemical equivalent of a dramatic plot twist.
The Awkward Intermediate
Suddenly, we have this funky intermediate molecule. Griff is now attached to the carbon of CO2, which still has one oxygen attached with a double bond, and the other oxygen is hanging out with a negative charge. It’s not pretty, but it’s necessary.
This intermediate is unstable and ready for the next act. Imagine a wobbly tower of LEGO bricks just waiting for the final piece.
Protonation – The Final Flourish
Here’s where the magic really happens. We add some acid, like hydrochloric acid (HCl). In our kitchen analogy, it’s like adding lemon juice to bring out the flavors. This step is called protonation.
The negatively charged oxygen atom on our intermediate molecule sees the positive hydrogen ion (proton) from the acid and goes wild! It snatches it up like a lifeline.
Poof! The negatively charged oxygen is neutralized and becomes a hydroxyl group (-OH). This transforms the molecule into a carboxylic acid. And this whole process is called carboxylation!
From Griff to Gourmet
So, what have we achieved? We started with a Grignard reagent, Griff, who was itching for a reaction. We introduced him to CO2, and through a series of atomic hugs and a dash of acid, we ended up with a carboxylic acid.

Carboxylic acids are incredibly important. They're found in everything from vinegar (acetic acid) to fats and proteins. They're the building blocks of much of the organic world.
The carboxylation of a Grignard reagent is like turning a simple, reactive ingredient into a complex, flavorful dish. It’s a fundamental reaction in organic chemistry, allowing chemists to build larger, more complex molecules.
Why This Matters – The Humble Beginnings
You might be thinking, "Okay, that's interesting, but why should I care?" Well, this reaction allows scientists to create molecules that wouldn't be easily accessible otherwise. It's like discovering a new spice that completely revolutionizes cooking.
Imagine trying to synthesize a specific drug without being able to add a carboxylic acid group where you need it. It would be like trying to bake a cake without flour!
The Grignard reagent's ability to grab onto CO2 and form a carboxylic acid is a powerful tool in organic synthesis. It's a testament to the elegance and efficiency of chemical reactions.

Grignard Reagents: More Than Just a Name
The discovery of Grignard reagents by Victor Grignard himself was a monumental achievement. It earned him the Nobel Prize in Chemistry in 1912. He was basically the rockstar chef of his day!
His invention opened up a whole new world of possibilities for organic chemists. It's like giving a painter a brand new set of colors to work with.
So next time you hear about a Grignard reagent, remember our little chef, Griff, who loves CO2 so much. It's a heartwarming story of atomic attraction and the power of chemical reactions to create something new and useful.
Beyond the Lab: Real-World Applications
Think about creating new polymers with specific properties or designing drugs that target diseases more effectively. Carboxylation of Grignard reagents plays a vital role in achieving these goals.
This reaction isn't just confined to the lab; it has real-world applications that impact our lives in countless ways. It's the unsung hero of many advancements in medicine, materials science, and more.
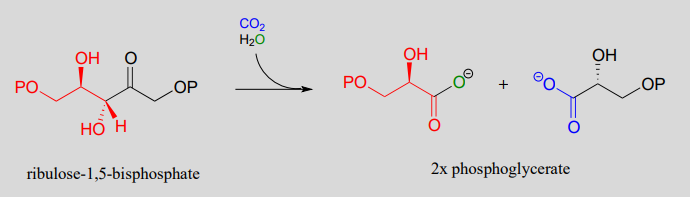
From developing sustainable plastics to creating novel pharmaceuticals, the possibilities are endless. It all starts with that simple, yet powerful interaction between a Grignard reagent and carbon dioxide.
The Grignard Legacy: A Continuing Story
The story of Grignard reagents and their ability to carboxylate is far from over. Scientists are constantly finding new ways to use these reactive compounds to create innovative materials and solutions.
Just as cooking evolves with new techniques and ingredients, so too does chemistry with the ongoing exploration of reactions like this. It is a dynamic process, full of surprises and discoveries.
So, the next time you think of organic chemistry as some obscure, complicated subject, remember Griff and his love for CO2. It's a story of simple ingredients, a little bit of magic, and the creation of something truly remarkable.
A Final Thought
Even the most complex scientific concepts can be understood and appreciated with a little imagination. So, embrace your inner chemist and explore the wonderful world of molecules!
It is a universe of tiny interactions which is the key to understanding the macro world.